Types of research studies5/15/2023 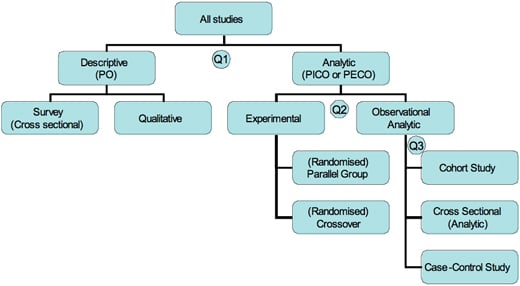
In order to obtain comprehensive data, research studies require diverse participants. Representation: Importance of Diversity in Clinical Research StudiesĬlinical studies allow scientists and researchers to answer specific questions about the effectiveness and safety of drugs, vaccines, devices, and treatments. If someone decides they no longer want to participate in the study, they can withdraw at any time. 
It is a process that continues with ongoing conversations between the research team and participants before, during, and after research participation. The process of informed consent does not end when someone signs the informed consent document. Participants should feel comfortable asking questions and discussing with the research team any concerns they might have. It is the responsibility of the research staff to help participants understand the information about the study and give them time to decide if they want to participate. If someone is interested in participating in a study, the research team may provide an informed consent document that includes details about the study, such as its purpose, how long it lasts, required procedures, potential risks, and whom to contact with questions or issues. It is the process of learning the key facts about the clinical study before deciding whether to participate. 
Informed consent is an essential part of participating in a clinical study.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
